Blog
Prescription Drug Abuse Among Pregnant Women:
http://rockcenter.msnbc.msn.com/_news/2012/07/05/12570381-prescription-drug-addiction-among-pregnant-women-becoming-monstrous-tidal-wave?lite
Addicted Mom on Baby’s Withdrawl; It’s All My Fault:
http://www.msnbc.msn.com/id/21134540/vp/48085618#48085618
Prescription Pill Epidemic Impacts Babies:
http://www.msnbc.msn.com/id/21134540/vp/48081187#48081187
News You Can Use:
Bill Banning Synthetic Drugs Passes Senate, Awaits President’s Signature
By Join Together Staff | June 29, 2012 | reprinted from: http://www.drugfree.org/join-together/drugs/bill-banning-synthetic-drugs-passes-senate-awaits-president%E2%80%99s-signature
The U.S. Senate passed a bill outlawing synthetic drugs this week, and awaits President Obama’s signature, The Cleveland Plain Dealer reports. The Synthetic Drug Abuse Prevention Act of 2012 bans drugs such as “bath salts” and synthetic marijuana at the federal level.
According to a news release from the office of Senator Charles Schumer of New York, the amendment included three bills relating to synthetic substances: one for bath salts, one for synthetic marijuana, and one for synthetic hallucinogens. Click on link above for more information.
USDTL Unveils New “bath Salts” Assay in Umbilical Cord</p>

Designer stimulants produce an euphoric high and have been described by the CDC as “a serious public threat.” These stimulants are packaged as bath salts, plant food or stain remover with labels that indicate “not for human consumption”. (CDC, 2011; Dargan et al, 2010; Dargan et al, 2011). Officials say the powdery substance mimics the effects of cocaine, ecstasy and LSD. These chemicals can cause hallucinations, paranoia, rapid heart rates, violent behavior and suicidal thoughts.
Healthcare workers are seeing neonatal abstinence syndrome (NAS) like symptoms similar to those seen in newborns exposed to the drugs listed above but cannot be attributed to opiates or other commonly screened for substances of abuse. Due in part to the lack of a widely-available, objective screen to identify newborns that have been exposed to designer stimulants, currently no specific research exists concerning the short-term or long-term harm caused by in utero exposure to designer stimulants. That said, the UK Teratology Information Service has warned that use of designer stimulants should be avoided during pregnancy (UKTIS, 2010).
CordStat® is an ideal clinical assay for healthcare practitioners concerned with monitoring newborns at high risk for having been exposed to Bath Salts/Designer Stimulants. The assay, which tests for 8 of the most common ingredients, can be ordered as an add-on test to one of our standard CordStat® 5-, 7-, 9-, 12- and 13-drug panels. To order, researchers and clinicians can contact Client Services at (800) 235-2367 or at clientservices@usdtl.com.
Established in 1991 as a specialty drug testing facility, USDTL quickly became a leader in the industry.USDTL has made significant breakthroughs by developing procedures to effectively use umbilical cord tissue and meconium specimens to diagnose alcohol misuse and substances of abuse. To find out more, please visit https://www.usdtl.com/.
Contact:
Bob Demaree, Clinical Projects Manager
bob.demaree@usdtl.com
(800) 235-2367
USDTL Now GSA Certified Contractor
DES PLAINES, Ill. (May 25, 2012) – Des Plaines, IL,—We are pleased to announce that United States Drug Testing Laboratories, Inc. (USDTL) has been awarded a 5-year Federal Supply Schedule (FSS) contract (V797P-2097D) from the Department of Veteran Affairs as part of the General Services Administration (GSA) Multiple Award Schedule which went into effect February 15, 2012. USDTL received the award for Schedule 621II Medical Laboratory Testing and Analytical Services as a certified small business enterprise.
VA Centers around the nation can now utilize USDTL’s state-of-the-art, long-term alcohol and drug biomarkers to assist with the treatment of chronic substance abuse. The contract also allows OB/GYN practitioners at all US military hospitals access to USDTL’s state-of-the-art toxicology services for newborn meconium and umbilical cord tissue testing for prenatal exposure to substances of abuse and heavy alcohol use.
USDTL is a leader in the toxicology industry; bringing new and innovative services to market to assist healthcare professionals to solve complex addiction issues. Government purchasers may contact Client Services at (800) 235-2367 or clientservice@usdtl.com to obtain more information and to coordinate specimen collection instructions and supplies.
Contact:
Joseph Jones, Vice President of Laboratory Services
joe.jones@usdtl.com
(800) 235-2367

Spring Break Ritual Could Lead to Brain Damage http://bit.ly/y8olhB
War Veterans with Mental Health Diagnoses More Likely to Receive Prescription Opioids for Pain http://bit.ly/wCFxPi
Wyoming kidney failure outbreak linked to “Spice” drug http://reut.rs/wWgjsG
Hawkins County Sheriff’s Office raids stores accused of selling synthetic drugs: Hawkins County Sheriff Ronnie L… http://bit.ly/Arv8oZ
Huffington Post guest columnist Michael Friedman, LMSW, reviews the signs of prescription painkiller misuse. http://fb.me/HAowMmdv
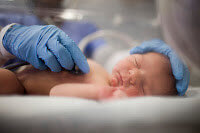
The Multidisciplinary Counterdrug Task Force Training: A Partnership Between the Florida National Guard and St. Petersburg College has developed a broadcast focusing on drug addicted infants.
Every day, babies are born addicted to prescription medications. They scream and twitch as their tiny bodies withdraw from the drugs their mothers took during pregnancy. All around the country, this problem grows as the number of prescription addicts rises. What can be done?
The broadcast is one hour long and is called Addicted at Birth. See how doctors, nurses, and other professionals treat these tiny addicts and why it is so expensive and time-consuming. You’ll learn why some pregnant women do not seek help during their pregnancy and why abruptly stopping prescription use can be harmful for both mother and the baby. Hear how law enforcement and other emergency responders play a big role in insuring help for the newborns.
Key Concepts:
- Understand the scope of the problem
- Learn how prescription-addicted babies are treated
- Understand why some pregnant women do not seek help
- See how law enforcement and emergency responders play a role in protecting these babies
For more information or to view the broadcast, please click here.
http://www.mctft.com/telecasts/view_course.aspx?telecastID=2012-03-29-1
Michael Sucher, MD
Medical Director, President and Contract Manager, Greenburg and Sucher PC.
http://www.youtube.com/watch?v=Kjxer0YWvRg
 Bath Salts – This terrible and relatively new type of drug is a growing problem across the United States. Here are some links to find out more:
Bath Salts – This terrible and relatively new type of drug is a growing problem across the United States. Here are some links to find out more:
From NIDA:
https://nida.nih.gov/research-topics/synthetic-cathinones-bath-salts
- Forensic vs. Clinical Drug Testing: Why Flexibility Matters for Your Organization
- USDTL’s Integration and Partnership With CourtFact
- New Year, New Capabilities: Offering Forensic & Clinical Testing Options
- Weather Delay
- The Detection of Delta-9-tetrahydrocannabinol, Delta-8-tetrahydrocannabinol, Delta-10-tetrahydrocannabinol, and Cannabidiol in Hair Specimens
- Umbilical Cord Tissue Testing for Ketamine
- Drugs of Abuse: A DEA Resource Guide (2024)
- Beyond THC and CBD: Understanding New Cannabinoids
- February 2026 (1)
- January 2026 (3)
- October 2025 (1)
- July 2025 (3)
- May 2025 (2)
- April 2025 (2)


