Meconium Collection: Nothing More, Nothing Less
Showing: Inc.
What you need to know about meconium collection.
by Michelle Lach, MSIMC
Meconium is the first stool of a newborn infant. It is produced in utero and consists of materials such as epithelial cells, bile, mucous, and more. In most newborns, meconium is generally passed in the first day or so of life, has no odor, and appears as a very dark, tar-like substance. This helps distinguish meconium from the next phase of passage called transitional stool.
Transitional stool will start to have an odor and present with a more brown, green, or yellow color as the newborn starts digesting milk. When drug testing the meconium of a newborn, it is important to note this difference since only meconium is created during gestation and transitional stool is created after birth. Collection of any stool other than meconium for drug testing purposes may result in a rejected specimen.
Unlike umbilical cord tissue, drugs are not distributed uniformly throughout the meconium specimen (see Figure 1). Because of this, the collection of the entire mass of meconium is highly encouraged to assure that there will be enough specimen to test, and that the maximum window of drug detection is achieved. It can take multiple passages of meconium before the newborn begins the transitional stool phase.
We require a minimum of 3 grams of meconium to be able to properly run our tests, so collecting the entire passage of meconium from newborns that have been exposed to substances of abuse is highly critical since they tend to have lower birth weights and create less specimen in the first place. If there is not enough specimen to run the test, the results are reported out as QNS. Quantity Not Sufficient (QNS) is a result of not having a sufficient quantity (volume) of specimen to test for the panels ordered.


Neonatal Drug Withdrawal
By Freepik© Studio
Maternal Nonnarcotic Drugs that cause neonatal psychomotor behavior consistent with withdrawal.
The Onset of Newborn Withdrawal Symptoms is Highly Variable
| Drug | Onset of Signs |
| Diazepam | Hours to Weeks |
| Alcohol | 3-12 Hours |
| Heroin | 24 Hours |
| Sedatives | 1-3 Days |
| Methadone | 1-7 Days |
| Opiates | 1-7 Days |
| Barbiturates | 1-14 Days |
– Click here to download the pdf.
Read an excerpt from the article Neonatal Drug Withdrawal below:
Signs characteristic of neonatal withdrawal have been attributed to intrauterine exposure to a variety of drugs. Other drugs cause signs in neonates because of acute toxicity. Chronic in utero exposure to a drug (eg, alcohol) can lead to permanent phenotypical and/or neurodevelopmental-behavioral abnormalities consistent with drug effect. Signs and symptoms of withdrawal worsen as drug levels decrease, whereas signs and symptoms of acute toxicity abate with drug elimination. Clinically important neonatal withdrawal most commonly results from intrauterine opioid exposure. The constellation of clinical findings associated with opioid withdrawal has been termed the neonatal abstinence syndrome (NAS). Among neonates exposed to opioids in utero, withdrawal signs will develop in 55% to 94%.1,2 Neonatal withdrawal signs have also been described in infants exposed antenatally to benzodiazepines,3,4 barbiturates,5,6 and alcohol.7,8
— Neonatal Drug Withdrawal https://doi.org/10.1542/peds.2011-3212
References:
-
Harper RG, Solish GI, Purow HM, Sang E, Panepinto WC. The effect of a methadone treatment program upon pregnant heroin addicts and their newborn infants. Pediatrics. 1974 ; 54 (3): 300–305 [PubMed]
-
Ostrea EM, Chavez CJ, Strauss ME. A study of factors that influence the severity of neonatal narcotic withdrawal. J Pediatr. 1976; 88 (4 pt 1): 642–645 [PubMed]
-
Rementería JL, Bhatt K. Withdrawal symptoms in neonates from intrauterine exposure to diazepam. J Pediatr. 1977; 90 (1): 123–126 [PubMed]
-
Athinarayanan P, Piero SH, Nigam SK, Glass L. Chloriazepoxide withdrawal in the neonate. Am J Obstet Gynecol. 1976; 124 (2): 212–213 [PubMed]
-
Bleyer WA, Marshall RE. Barbiturate withdrawal syndrome in a passively addicted infant. JAMA. 1972; 221 (2): 185–186 [PubMed]
- Desmond MM, Schwanecke RP, Wilson GS, Yasunaga S, Burgdorff I. Maternal barbiturate utilization and neonatal withdrawal symptomatology. J Pediatr. 1972; 80 (2): 190–197 [PubMed]
- Pierog S, Chandavasu O, Wexler I. Withdrawal symptoms in infants with the fetal alcohol syndrome. J Pediatr. 1977; 90 (4): 630–633 [PubMed]
- Nichols MM. Acute alcohol withdrawal syndrome in a newborn. Am J Dis Child. 1967; 113 (6): 714–715 [PubMed]

What You Need To Know: Testing for Drug Exposure vs. Ingestion
Testing for environmental exposure to illicit drugs is a powerful tool for protecting the welfare of children. Exposure testing is different from typical drug testing, and when properly done, has the potential to reduce the risk of harm to children.
No Metabolite Does NOT Mean No Exposure
Testing labs often apply government workplace testing guidelines to child exposure testing samples. Under workplace guidelines, negative results are reported when drug metabolites are absent in the testing sample, even if the native drug is present.
Child hair and nail samples for exposure testing often do not contain drug metabolites because the child has not ingested illicit substances. Adhering to workplace guidelines can result in false negative reporting for drug exposure, especially when children are involved.
Environmental Exposure
Environmental Exposure testing is most effective in alternative sample types, such as hair and fingernails. For example, hair testing is 3.5x more likely to detect methamphetamine exposure than urine testing. Typical drug testing samples are washed to remove drug biomarkers resulting from exposure. Environmental exposure testing eliminates this step.
References:
- Kelleher, K., Chaffin, M., Hollenberg, J., & Fischer, E. (1994). Alcohol and drug disorders among physically abusive and neglectful parents in a community-based sample. American Journal of Public Health, 84(10), 1586-1590.
- Murphy, J. M., Jellinek, M., Quinn, D., Smith, G., Poitrast, F. G., & Goshko, M. (1991). Substance abuse and serious child mistreatment: Prevalence, risk, and outcome in a court sample. Child abuse & neglect, 15(3), 197-211.
– Click here to download the pdf.
When a child is exposed to illegal substance abuse they often also face other coexisting obstacles to a normal life – neglect, abuse, violence, and other vulnerabilities. Substance abuse is a disease, one that often prevents adults from doing what is in a child’s best interests. Our environmental exposure test for children can help.
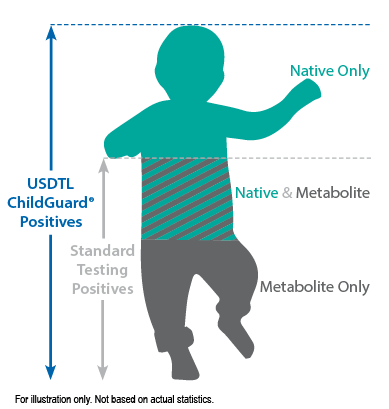
Our hair environmental exposure test is the only drug test designed to detect passive exposure to drugs and detect both native drugs and drug metabolites in the hair specimen. Drug metabolites are produced in the body only if drugs have been ingested. Children in drug exposed environments are most often not drug users themselves, so drug metabolites are typically absent in child specimens. However, the hair, like a sponge, can absorb non-metabolized drug (native drug) if it is exposed through things such as touching or being in contact with drugs or drug users.
Standard hair tests with other labs will only report a positive exposure result if drug metabolites are detected, even when the native drug is in the child’s hair specimen. Our hair environmental exposure test reports a positive result if either native drugs or drug metabolites are detected.
A hair exposure test can provide evidence of drugs in a child’s environment for the past 3 months. A positive test result suggests that the child has experienced one or more of the following: passive inhalation of drug smoke, contact with drug smoke, contact with sweat or sebum (skin oil) of a drug user, contact with the actual drug, or accidental or intentional ingestion of illegal drugs.
ChildGuard®is the only child hair test designed to detect exposure to native drugs and drug metabolites.

By Joseph Salerno
The movement and location of physical evidence from the time it is obtained until the time it is presented in court is the legal definition of chain of custody. The results of any newborn alcohol or substance of abuse test performed at USDTL may eventually be presented as evidence in a court of law, and this is why USDTL maintains universal chain of custody regardless of the client source of testing specimens. A court can exclude the results of a test if a chain of custody for the newborn sample was not maintained by the hospital and USDTL.
Chain of custody for specimens sent to USDTL is maintained as a chronological paper trail of collection and transfers of specimens throughout the testing process. The paper trail is signed and dated by each person who handles the specimen, both when they receive the specimen into their own hands, and when they hand it off to the next person in the process. Less transfers of a specimen that need to be documented is better for the chain of custody overall. A well maintained and legal chain of custody begins at the time of specimen collection and continues uninterrupted until test results have been presented in court, if necessary.
There are several key elements of the chain of custody for alcohol and drug test samples that must be present when samples arrive at USDTL. First, the specimen container must be sealed with an intact security seal. Next, the sample must be accompanied by a Chain of Custody and Control Form with an identification number matching the number on the specimen container. The Chain of Custody and Control Form is the first piece of the chain of custody paper trail. Thirdly, the Chain of Custody and Control Form must be signed and dated by an authorized agent from the client. If one or more of these elements are missing, USDTL must return the sample to the client.
An unbroken chain of custody ensures sample integrity in several ways that preserve the legal usefulness of alcohol and drug testing results.
Chain of custody ensures that the original sample is the same as the one that is tested and ensures that the integrity of the sample is preserved during transport. Tampering, substitution, or alteration of the sample prior to being tested is prevented by the chain of custody process, which ensures thatit has been handled only by the donor, a qualified collector, and lab testing personnel.
Maintaining chain of custody for newborn samples destined for alcohol and drug testing is a simple process, but all those who handle a drug testing specimen need to be vigilant about the process nonetheless. Diligent maintenance of chain of custody is always in the child’s best interest. Unfortunately, it is only when the legal impact of an improperly maintained chain of custody is realized, that the full value of a well maintained chain of custody is understood. Ultimately, chain of custody protects the institution that is collecting the specimen, as well as the newborn whose health and well-being may rely on the results of a USDTL alcohol or drug test.
Reference: Giannelli, P. (1996). Forensic Science: Chain of Custody. Criminal Law Bulletin, 32(5), 447-465.
Dear Valued Client,
We are proud to announce that we are the first laboratory in the world to be ISO/IEC 17025 accredited for drug and alcohol testing in umbilical cord, fingernail, and toenail specimens. On September 4, 2015, USDTL attained ISO/IEC 17025 accreditation showing full compliance with the international testing standards. We have received our accreditation from ANSI-ASQ National Accreditation Board, demonstrating technical competence in the field of forensic testing. The scope of our ISO/IEC 17025 accreditation encompasses all specimen types and methods of analysis utilized in our laboratory.
Our laboratory has always maintained this level of quality and competency since our humble beginnings in 1991, bringing our clients the most responsive, personal service in the drug and alcohol testing industry. ISO/IEC 17025 accreditation reaffirms that commitment to our clients, for all aspects of our testing and client advocacy. You can always have absolute confidence that the results of every specimen tested by our laboratory will meet the highest of international standards.
ISO/IEC 17025 is the single most important standard applied to testing and calibration laboratories around the globe. Laboratories accredited to the ISO/IEC 17025 standard have demonstrated that they are technically competent and able to reproducibly generate accurate, precise and consistent data.
The practical benefits for clients of USDTL of ISO/IEC 17025 accreditation are seen on a continual basis:
- Continuously produced testing results of the highest quality, validity, and integrity;
- Improved customer communication and resolution of customer issues;
- Continual improvement of our management system, with an emphasis on the responsibilities of senior management;
- Fast resolution of laboratory issues regarding methods and equipment.
- Evidential acceptance of USDTL laboratory results in virtually all jurisdictions.
To view our certificate of accreditation by ANAB 17025:2017 Forensic Science Testing and Calibration Lab. If you have questions about ISO/IEC 17025 accreditation, please contact us at clientservices@usdtl.com or 800.235.2367.
Sincerely,
Adam Negrusz, Ph.D., F-ABFT
Laboratory Director
- Forensic vs. Clinical Drug Testing: Why Flexibility Matters for Your Organization
- USDTL’s Integration and Partnership With CourtFact
- New Year, New Capabilities: Offering Forensic & Clinical Testing Options
- Weather Delay
- The Detection of Delta-9-tetrahydrocannabinol, Delta-8-tetrahydrocannabinol, Delta-10-tetrahydrocannabinol, and Cannabidiol in Hair Specimens
- Umbilical Cord Tissue Testing for Ketamine
- Drugs of Abuse: A DEA Resource Guide (2024)
- Beyond THC and CBD: Understanding New Cannabinoids
- February 2026 (1)
- January 2026 (3)
- October 2025 (1)
- July 2025 (3)
- May 2025 (2)
- April 2025 (2)