Marijuana: A Multifaceted Movement
Showing: United States Drug Testing Laboratories

In 1996, California became the first state to pass legislation condoning the use of marijuana for medicinal purposes, and since the onset of that law, a powerful trend was set. Over 33 other states, including the District of Columbia, have now adopted various statutes for the permittance of recreational and medicinal marijuana. Although many government entities have sided that the benefits of the cannabis plant outweigh the risks, others are anxious that the ease of accessibility may cause an influx of misleading notions regarding the plant. Its exposure to vulnerable populations, including adolescents, those pregnant, or individuals suffering from preexisting psychiatric disorders continues to be a significant concern among communities.
The Impact of Legalization on Local Communities
In a cross-sectional marijuana dispensary density study from 2001-2012 in California, associations between marijuana abuse/dependence hospitalizations disclosed that an additional one dispensary per square mile was associated with a 6.8% increase in the number of marijuana hospitalizations. The study’s findings concluded that increased availability of marijuana in zip codes with a higher density of dispensaries continues to be a probable correlation to the increased hospitalizations in dispensary-dense areas.1
Despite this study and others, a recent CBS News Poll found that support for marijuana legalization has risen among groups that have historically opposed it. More than half of Republicans (56 percent) now think marijuana use should be legal due to reasons such as marijuana being less harmful than alcohol and believing it is less harmful than other drugs.2 However, increases in marijuana potency is triggering a valid fear that the levels of THC in today’s plants are more toxic than therapeutic.
Increased Potency, Increased Risks
As highly potent cannabis increases in availability, scientists who study marijuana and the effects it has to the human body are becoming disturbed with the increasingly high rates of potency in delta-9-tetrahydrocannabinol (THC)–the main compound responsible for the drug’s psychoactive effects. According to a U.S. Drug Enforcement Administration seize, the potency of marijuana has increased from about 4% THC in 1995 to about 12% in 2014. By 2017 marijuana samples were up to 17.1% THC, totaling an increase of more than 300% from 1995-2017. Concentrated cannabis products known as hash and hash oil are also reaching potency levels as high as 80-90% THC.3
Nora Volkow, Director of the National Institute on Drug Abuse (NIDA) states, “The notion that it is a completely safe drug is incorrect when you start to address the consequences of this very high content of Delta-9-THC.”
The levels of THC within cannabis is imperative when factoring the effects it can have on the body when consumed. Low THC levels have been known to have less adverse effects compared to high THC levels.
Low THC Content:
- Decreases Anxiety
- Treats Nausea
- Relaxation3
High THC Content:
- Panic Attacks
- Psychosis
- Paranoia
- Cannabinoid Hyperemesis Syndrome3
The Diagnostic and Statistical Manual of Mental Disorders (DSM-5) of the American Psychiatric Association now includes Cannabis Use Disorder (CUD) as a substance use disorder (SUD) diagnosis. Not all cannabis users develop CUD, however it is becoming more common than we think and can be serious. Normalizing use and reducing perception of harm can increase the development of CUD.4
DSM-5 Cannabis Withdrawal Symptoms:
- Anxiety, restlessness
- Depression, irritability
- Insomnia/odd dreams
- Physical symptoms, e.g.Tremors
- Decreased appetite4
In a longitudinal study published in Addiction, CUD was found to be significantly associated with psychotic and depressive symptoms.5
The Association Between Cannabis Use and Psychiatric Comorbidity
Cannabis use is recognized as a contributing factor for developing a psychotic disorder, children and teens with a family history of psychosis are most vulnerable.4
In a long-term prospective study, 1265 children born in Christchurch, New Zealand in 1977 were assessed repeatedly for psychosis symptoms due to daily exposure of cannabis in utero, which contributed to psychotic symptoms portrayed in these children at between the ages of 18-25. There was a significant correlation between cannabis use and later development of psychosis.4
A study conducted by Lancet Psychiatry found that three European cities–London, Paris, and Amsterdam, where high-potency weed is most prevalent, also have the highest rates of new cases of psychosis. The study indicates that daily pot users are three times more likely to endure a psychotic episode compared to an individual who has abstained from the substance.4
High potency forms of marijuana known as wax, butane hash oil, dabs, or shatter are growing in popularity and are more likely to induce psychotic states. The principal psychoactive component of cannabis is THC, which binds to cannabinoid-1 (CB-1) receptors found throughout the central nervous system. Studies specify that pure THC and CB1 agonists can produce psychotic symptoms including suspiciousness, paranoia, thought disorganization, and derealization.7
Marta Di Forti, lead author from the Institute of Psychiatry, Psychology, and Neuroscience at King’s College London says, “As the legal status of cannabis change in many countries and states, and as we consider the medicinal properties of some types of cannabis, it is of vital public health importance that we also consider the potential adverse effects that are associated with daily cannabis use, especially high potency varieties.”6
Maternal Marijuana Use
The adverse effects of marijuana can become extremely dangerous when the substance is used among those pregnant. According to the National Survey on Drug Use and Health, nearly 4% of pregnant women in 2007 and 2012 used marijuana in the past 30 days. Long-term neurobehavioral studies have shown that negative consequences have been found in children exposed to marijuana in utero such as altered neural functioning, behavioral deficits, emotional deficits, low academic achievement, and increased risk of adolescent substance use initiation.8
The uptick of marijuana legalization has generated a significant concern among obstetricians, gynecologists, and neonatal practitioners who are combating misleading claims that marijuana use during pregnancy is safe. According to the Center for Disease Control and Prevention (CDC), about 1 in 25 women in the U.S. report using marijuana while pregnant, despite the fact that marijuana use during pregnancy may increase the baby’s risk of developmental problems and low birth weight.9 The American College of Obstetricians and Gynecologists (ACOG) recommends that obstetrician-gynecologists counsel women against using marijuana while trying to get pregnant, during pregnancy, and while breastfeeding.10 Studies have found that cannabinoid receptors appear in the fetal brain around the 14th week of gestation and are located in areas where cognitive and behavioral functioning develop.11
According to a qualitative study, women reported that although they were consistently seeking prenatal care throughout their pregnancy, information and resources regarding maternal marijuana use was either not helpful or non-existent, resulting in the assumption that marijuana did not pose a significant threat to a developing fetus.12 The study concludes that absenteeism of perinatal marijuana education can lead to an increase of use among pregnant women.12
Testing for Abuse
Cannabis is not a harmless substance. It has been found to have addictive properties, which can lead to impairments and cause serious health risks. Our tests are designed to identify the detection of short-term and long-term marijuana usage. Each available specimen type provides a unique window of detection.
- Hair: Up to approximately 3 months prior to collection.
- Nail: Up to approximately 3-6 months prior to collection.
- Umbilical Cord: Up to approximately 20 weeks prior to birth.
- Meconium: Up to approximately 20 weeks prior to birth.
- Urine: Up to approximately 2-3 days prior to collection.
We believe that to remain at the forefront of toxicology, it is imperative to offer testing services for all substances that may pose an increased risk for abuse and dependence. Our continued investment in developing and implementing testing for drug ingestion and exposure helps us address substances that most concern you.
References:
- Mair, C., Freisthler, B., Ponicki, W. R., & Gaidus, A. (2015, September 01). “The impacts of marijuana dispensary density and neighborhood ecology on marijuana abuse and dependence.” Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4536157/
- “Support for marijuana legalization hits new high, CBS News poll finds.” (n.d.). Retrieved from https://www.cbsnews.com/news/support-for-marijuana-legalization-hits-new-high-cbs-news-poll-finds/
- Chatterjee, R. (2019, May 15). “Highly Potent Weed Has Swept The Market, Raising Concerns About Health Risks.” Retrieved from https://www.npr.org/sections/health-shots/2019/05/15/723656629/highly-potent-weed-has-swept-the-market-raising-concerns-about-health-risks
- Hasin, D. S. (2018, January). “US Epidemiology of Cannabis Use and Associated Problems.” Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5719106/
- Pond, E. (2019, January 28). “Cannabis Use, Cannabis Use Disorder Linked to Psychotic, Depressive Symptoms.” Retrieved from https://www.psychiatryadvisor.com/home/topics/addiction/cannabis-use-cannabis-use-disorder-linked-to-psychotic-depressive-symptoms/
- Robinson, J. (2019, March 20). “Daily use of high-potency cannabis increases risk of psychosis by four times, study finds.” Retrieved from https://www.pharmaceutical-journal.com/news-and-analysis/news/daily-use-of-high-potency-cannabis-increases-risk-of-psychosis-by-four-times-study-finds/20206308.article?firstPass=false
- Corey J. Keller, Evan C. Chen, Kimberly Brodsky & Jong H. Yoon(2016)“A case of butane hash oil (marijuana wax)–induced psychosis, Substance Abuse”, 37:3, 384-386, DOI: 10.1080/08897077.2016.1141153
- Jones, J. (2018).“Medical Marijuana Laws and Maternal Marijuana Use.” Des Plaines, IL: Archives of Women Health and Care.
- “What You Need to Know About Marijuana Use and Pregnancy” | Fact Sheets | CDC. (n.d.). Retrieved from https://www.cdc.gov/marijuana/factsheets/pregnancy.htm
- National Institute on Drug Abuse. (n.d.).“Can marijuana use during and after pregnancy harm the baby?” Retrieved from https://www.drugabuse.gov/publications/research-reports/marijuana/can-marijuana-use-during-pregnancy-harm-baby
- Day, N. L., Goldschmidt, L., Day, R., Larkby, C., & Richardson, G. A. (2015, June). “Prenatal marijuana exposure, age of marijuana initiation, and the development of psychotic symptoms in young adults.” Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/25534593
- Jarlenski, M., Tarr, J. A., Holland, C. L., Farrell, D., & Chang, J. C. (2016). “Pregnant Women’s Access to Information About Perinatal Marijuana Use: A Qualitative Study.” Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/27131908
What you need to know about meconium collection.
by Michelle Lach, MSIMC
Meconium is the first stool of a newborn infant. It is produced in utero and consists of materials such as epithelial cells, bile, mucous, and more. In most newborns, meconium is generally passed in the first day or so of life, has no odor, and appears as a very dark, tar-like substance. This helps distinguish meconium from the next phase of passage called transitional stool.
Transitional stool will start to have an odor and present with a more brown, green, or yellow color as the newborn starts digesting milk. When drug testing the meconium of a newborn, it is important to note this difference since only meconium is created during gestation and transitional stool is created after birth. Collection of any stool other than meconium for drug testing purposes may result in a rejected specimen.
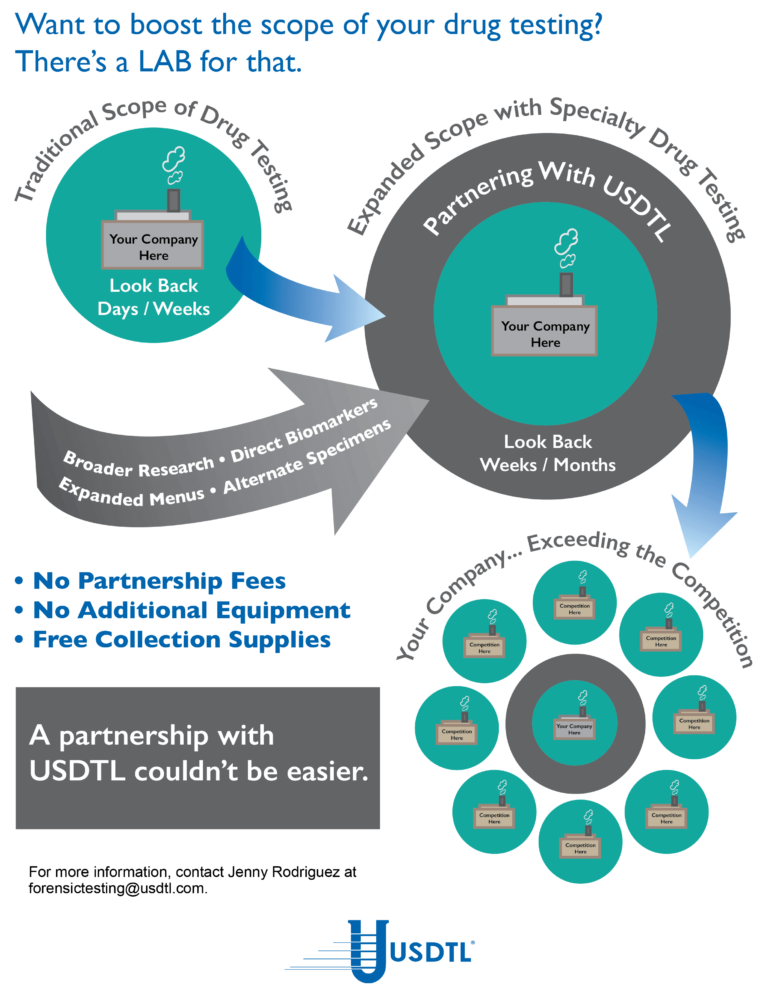
Unlike umbilical cord tissue, drugs are not distributed uniformly throughout the meconium specimen (see Figure 1). Because of this, the collection of the entire mass of meconium is highly encouraged to assure that there will be enough specimen to test, and that the maximum window of drug detection is achieved. It can take multiple passages of meconium before the newborn begins the transitional stool phase.
We require a minimum of 3 grams of meconium to be able to properly run our tests, so collecting the entire passage of meconium from newborns that have been exposed to substances of abuse is highly critical since they tend to have lower birth weights and create less specimen in the first place. If there is not enough specimen to run the test, the results are reported out as QNS. Quantity Not Sufficient (QNS) is a result of not having a sufficient quantity (volume) of specimen to test for the panels ordered.


Neonatal Drug Withdrawal
By Freepik© Studio
Maternal Nonnarcotic Drugs that cause neonatal psychomotor behavior consistent with withdrawal.
The Onset of Newborn Withdrawal Symptoms is Highly Variable
| Drug | Onset of Signs |
| Diazepam | Hours to Weeks |
| Alcohol | 3-12 Hours |
| Heroin | 24 Hours |
| Sedatives | 1-3 Days |
| Methadone | 1-7 Days |
| Opiates | 1-7 Days |
| Barbiturates | 1-14 Days |
– Click here to download the pdf.
Read an excerpt from the article Neonatal Drug Withdrawal below:
Signs characteristic of neonatal withdrawal have been attributed to intrauterine exposure to a variety of drugs. Other drugs cause signs in neonates because of acute toxicity. Chronic in utero exposure to a drug (eg, alcohol) can lead to permanent phenotypical and/or neurodevelopmental-behavioral abnormalities consistent with drug effect. Signs and symptoms of withdrawal worsen as drug levels decrease, whereas signs and symptoms of acute toxicity abate with drug elimination. Clinically important neonatal withdrawal most commonly results from intrauterine opioid exposure. The constellation of clinical findings associated with opioid withdrawal has been termed the neonatal abstinence syndrome (NAS). Among neonates exposed to opioids in utero, withdrawal signs will develop in 55% to 94%.1,2 Neonatal withdrawal signs have also been described in infants exposed antenatally to benzodiazepines,3,4 barbiturates,5,6 and alcohol.7,8
— Neonatal Drug Withdrawal https://doi.org/10.1542/peds.2011-3212
References:
-
Harper RG, Solish GI, Purow HM, Sang E, Panepinto WC. The effect of a methadone treatment program upon pregnant heroin addicts and their newborn infants. Pediatrics. 1974 ; 54 (3): 300–305 [PubMed]
-
Ostrea EM, Chavez CJ, Strauss ME. A study of factors that influence the severity of neonatal narcotic withdrawal. J Pediatr. 1976; 88 (4 pt 1): 642–645 [PubMed]
-
Rementería JL, Bhatt K. Withdrawal symptoms in neonates from intrauterine exposure to diazepam. J Pediatr. 1977; 90 (1): 123–126 [PubMed]
-
Athinarayanan P, Piero SH, Nigam SK, Glass L. Chloriazepoxide withdrawal in the neonate. Am J Obstet Gynecol. 1976; 124 (2): 212–213 [PubMed]
-
Bleyer WA, Marshall RE. Barbiturate withdrawal syndrome in a passively addicted infant. JAMA. 1972; 221 (2): 185–186 [PubMed]
- Desmond MM, Schwanecke RP, Wilson GS, Yasunaga S, Burgdorff I. Maternal barbiturate utilization and neonatal withdrawal symptomatology. J Pediatr. 1972; 80 (2): 190–197 [PubMed]
- Pierog S, Chandavasu O, Wexler I. Withdrawal symptoms in infants with the fetal alcohol syndrome. J Pediatr. 1977; 90 (4): 630–633 [PubMed]
- Nichols MM. Acute alcohol withdrawal syndrome in a newborn. Am J Dis Child. 1967; 113 (6): 714–715 [PubMed]

What You Need To Know: Testing for Drug Exposure vs. Ingestion
Testing for environmental exposure to illicit drugs is a powerful tool for protecting the welfare of children. Exposure testing is different from typical drug testing, and when properly done, has the potential to reduce the risk of harm to children.
No Metabolite Does NOT Mean No Exposure
Testing labs often apply government workplace testing guidelines to child exposure testing samples. Under workplace guidelines, negative results are reported when drug metabolites are absent in the testing sample, even if the native drug is present.
Child hair and nail samples for exposure testing often do not contain drug metabolites because the child has not ingested illicit substances. Adhering to workplace guidelines can result in false negative reporting for drug exposure, especially when children are involved.
Environmental Exposure
Environmental Exposure testing is most effective in alternative sample types, such as hair and fingernails. For example, hair testing is 3.5x more likely to detect methamphetamine exposure than urine testing. Typical drug testing samples are washed to remove drug biomarkers resulting from exposure. Environmental exposure testing eliminates this step.
References:
- Kelleher, K., Chaffin, M., Hollenberg, J., & Fischer, E. (1994). Alcohol and drug disorders among physically abusive and neglectful parents in a community-based sample. American Journal of Public Health, 84(10), 1586-1590.
- Murphy, J. M., Jellinek, M., Quinn, D., Smith, G., Poitrast, F. G., & Goshko, M. (1991). Substance abuse and serious child mistreatment: Prevalence, risk, and outcome in a court sample. Child abuse & neglect, 15(3), 197-211.
– Click here to download the pdf.
When a child is exposed to illegal substance abuse they often also face other coexisting obstacles to a normal life – neglect, abuse, violence, and other vulnerabilities. Substance abuse is a disease, one that often prevents adults from doing what is in a child’s best interests. Our environmental exposure test for children can help.
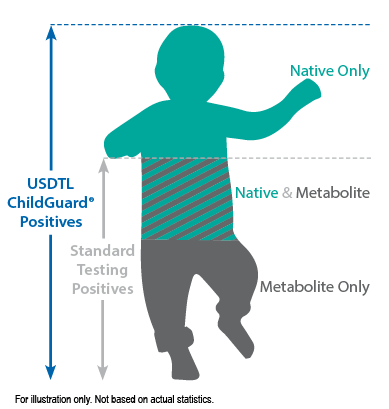
Our hair environmental exposure test is the only drug test designed to detect passive exposure to drugs and detect both native drugs and drug metabolites in the hair specimen. Drug metabolites are produced in the body only if drugs have been ingested. Children in drug exposed environments are most often not drug users themselves, so drug metabolites are typically absent in child specimens. However, the hair, like a sponge, can absorb non-metabolized drug (native drug) if it is exposed through things such as touching or being in contact with drugs or drug users.
Standard hair tests with other labs will only report a positive exposure result if drug metabolites are detected, even when the native drug is in the child’s hair specimen. Our hair environmental exposure test reports a positive result if either native drugs or drug metabolites are detected.
A hair exposure test can provide evidence of drugs in a child’s environment for the past 3 months. A positive test result suggests that the child has experienced one or more of the following: passive inhalation of drug smoke, contact with drug smoke, contact with sweat or sebum (skin oil) of a drug user, contact with the actual drug, or accidental or intentional ingestion of illegal drugs.
ChildGuard®is the only child hair test designed to detect exposure to native drugs and drug metabolites.

By Joseph Salerno
The movement and location of physical evidence from the time it is obtained until the time it is presented in court is the legal definition of chain of custody. The results of any newborn alcohol or substance of abuse test performed at USDTL may eventually be presented as evidence in a court of law, and this is why USDTL maintains universal chain of custody regardless of the client source of testing specimens. A court can exclude the results of a test if a chain of custody for the newborn sample was not maintained by the hospital and USDTL.
Chain of custody for specimens sent to USDTL is maintained as a chronological paper trail of collection and transfers of specimens throughout the testing process. The paper trail is signed and dated by each person who handles the specimen, both when they receive the specimen into their own hands, and when they hand it off to the next person in the process. Less transfers of a specimen that need to be documented is better for the chain of custody overall. A well maintained and legal chain of custody begins at the time of specimen collection and continues uninterrupted until test results have been presented in court, if necessary.
There are several key elements of the chain of custody for alcohol and drug test samples that must be present when samples arrive at USDTL. First, the specimen container must be sealed with an intact security seal. Next, the sample must be accompanied by a Chain of Custody and Control Form with an identification number matching the number on the specimen container. The Chain of Custody and Control Form is the first piece of the chain of custody paper trail. Thirdly, the Chain of Custody and Control Form must be signed and dated by an authorized agent from the client. If one or more of these elements are missing, USDTL must return the sample to the client.
An unbroken chain of custody ensures sample integrity in several ways that preserve the legal usefulness of alcohol and drug testing results.
Chain of custody ensures that the original sample is the same as the one that is tested and ensures that the integrity of the sample is preserved during transport. Tampering, substitution, or alteration of the sample prior to being tested is prevented by the chain of custody process, which ensures thatit has been handled only by the donor, a qualified collector, and lab testing personnel.
Maintaining chain of custody for newborn samples destined for alcohol and drug testing is a simple process, but all those who handle a drug testing specimen need to be vigilant about the process nonetheless. Diligent maintenance of chain of custody is always in the child’s best interest. Unfortunately, it is only when the legal impact of an improperly maintained chain of custody is realized, that the full value of a well maintained chain of custody is understood. Ultimately, chain of custody protects the institution that is collecting the specimen, as well as the newborn whose health and well-being may rely on the results of a USDTL alcohol or drug test.
Reference: Giannelli, P. (1996). Forensic Science: Chain of Custody. Criminal Law Bulletin, 32(5), 447-465.
- Forensic vs. Clinical Drug Testing: Why Flexibility Matters for Your Organization
- USDTL’s Integration and Partnership With CourtFact
- New Year, New Capabilities: Offering Forensic & Clinical Testing Options
- Weather Delay
- The Detection of Delta-9-tetrahydrocannabinol, Delta-8-tetrahydrocannabinol, Delta-10-tetrahydrocannabinol, and Cannabidiol in Hair Specimens
- Umbilical Cord Tissue Testing for Ketamine
- Drugs of Abuse: A DEA Resource Guide (2024)
- Beyond THC and CBD: Understanding New Cannabinoids
- February 2026 (1)
- January 2026 (3)
- October 2025 (1)
- July 2025 (3)
- May 2025 (2)
- April 2025 (2)