Urine Resources
Urine Resources
Urine Drug Testing Urine Drug Panels Urine Collection Instructions
Urine Videos
There are currently no Videos. Please check back later.
Urine Infographics
There are currently no Fingernail Infographics. Please check back later.
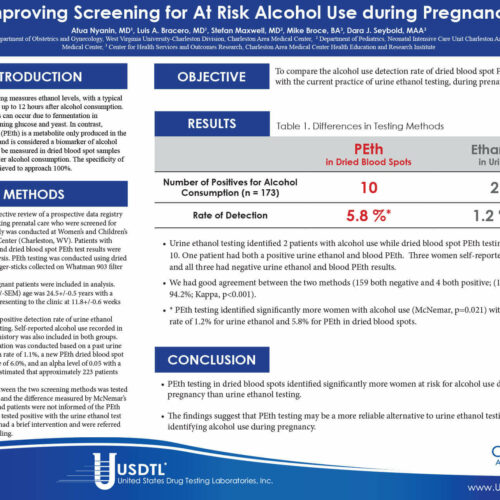
Urine Poster Presentations
Urine Articles
Clients Receive Custom Solutions with USDTL 01-Jan-2010
Urine Announcements
Psilocin and Bufotenine Testing in Urine
06-Jan-2025
We are announcing the addition of psilocin and bufotenine to our urine drug testing. These psychoactive substances can be ordered as an individual test or an add-on to any urine drug panel beginning January 6, 2025.
19-May-2025
We are excited to announce new offerings and improvements to our testing coming on May 19, 2025.
18-Apr-2016
Quantity Not Sufficient (QNS) Explained
02-Mar-2016
Cutoffs for Antihistamine Drugs in Urine Lowered
02-Aug-2015
USDTL Urine Research
USDTL Assisted Urine Research
Expressive and Receptive Language Functioning in Preschool Children with Prenatal Cocaine Exposure 01-Oct-2004
View All USDTL Assisted Research
Foundational Urine Research
View All Foundational Research
Urine White Papers
Psilocin: An Annotated Bibliography 25-Jun-2025
Urine FAQs
There are currently no FAQs. Please check back later.
Contact USDTL
1.800.235.2367
Client Services
Business Hours (CT)
Mon.-Fri. 7am-6pm
Sat. 8am-5pm
Holidays Closure
New Year's Day
Memorial Day
Independence Day
Labor Day
Thanksgiving Day
Christmas Day
Request Your Collection Supplies
For your convenience, USDTL provides test collection supplies at no additional charge.
Newsletters, Posters, and Catalogs
Our print materials will keep you up to date on the latest news in drug and alcohol testing.