Blog
By: Amy Racines, MS, Research & Development Senior Project Coordinator, USDTL
Benzodiazepines are a group of drugs which act as a central nervous system depressant and are used for the treatment of insomnia and anxiety. Although benzodiazepines are prescribed by doctors, they are also misused, most commonly in combination with opioids and alcohol. USDTL is excited to announce the addition of several benzodiazepines to the benzodiazepine panel in our umbilical cord assay beginning July 5th, 2023. Two of these added analytes are the designer benzodiazepines etizolam and flualprazolam, which can be purchased online.
The structures of medically approved benzodiazepines can be modified to create new compounds, commonly referred to as designer benzodiazepines, which are not medically approved. Etizolam and flualprazolam are two such designer benzodiazepines. Many times, these designer benzodiazepines are more potent than the medical benzodiazepines. Additionally, changing the structure to create these designer benzodiazepines affects the ability of this drug to be detected. Therefore, laboratories must update their methods to effectively detect new designer benzodiazepines.
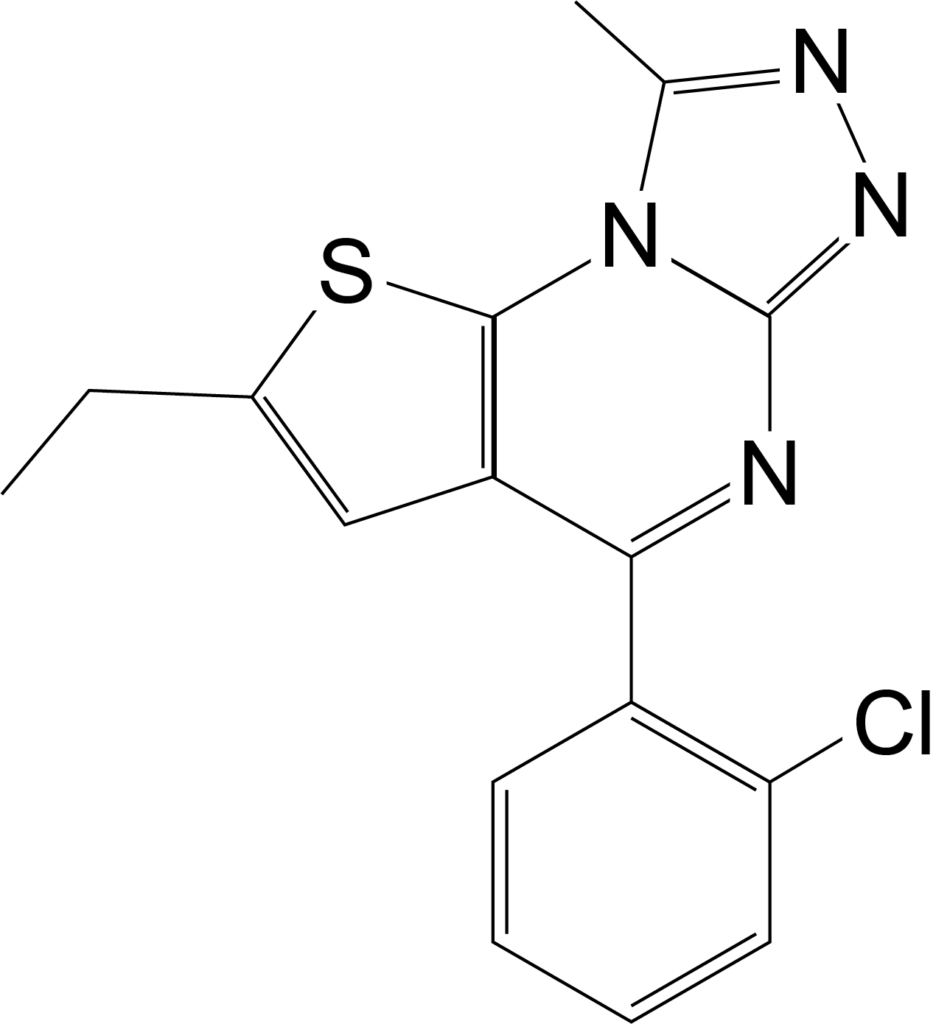 Etizolam has recently emerged in the illegal drug market in Europe and the United States, but it is an approved medication in Japan, India and Italy1. In 2018, the DEA reported 1,716 cases involving etizolam, but in 2021 that number rose to 4,252. Clinical studies have indicated that etizolam is about 10 times more potent than the medical benzodiazepine, diazepam. Common negative side effects of etizolam include drowsiness, sedation, muscle weakness and incoordination, fainting, headache, confusion, depression, slurred speech, visual disturbances, and changes in libido1.
Etizolam has recently emerged in the illegal drug market in Europe and the United States, but it is an approved medication in Japan, India and Italy1. In 2018, the DEA reported 1,716 cases involving etizolam, but in 2021 that number rose to 4,252. Clinical studies have indicated that etizolam is about 10 times more potent than the medical benzodiazepine, diazepam. Common negative side effects of etizolam include drowsiness, sedation, muscle weakness and incoordination, fainting, headache, confusion, depression, slurred speech, visual disturbances, and changes in libido1.
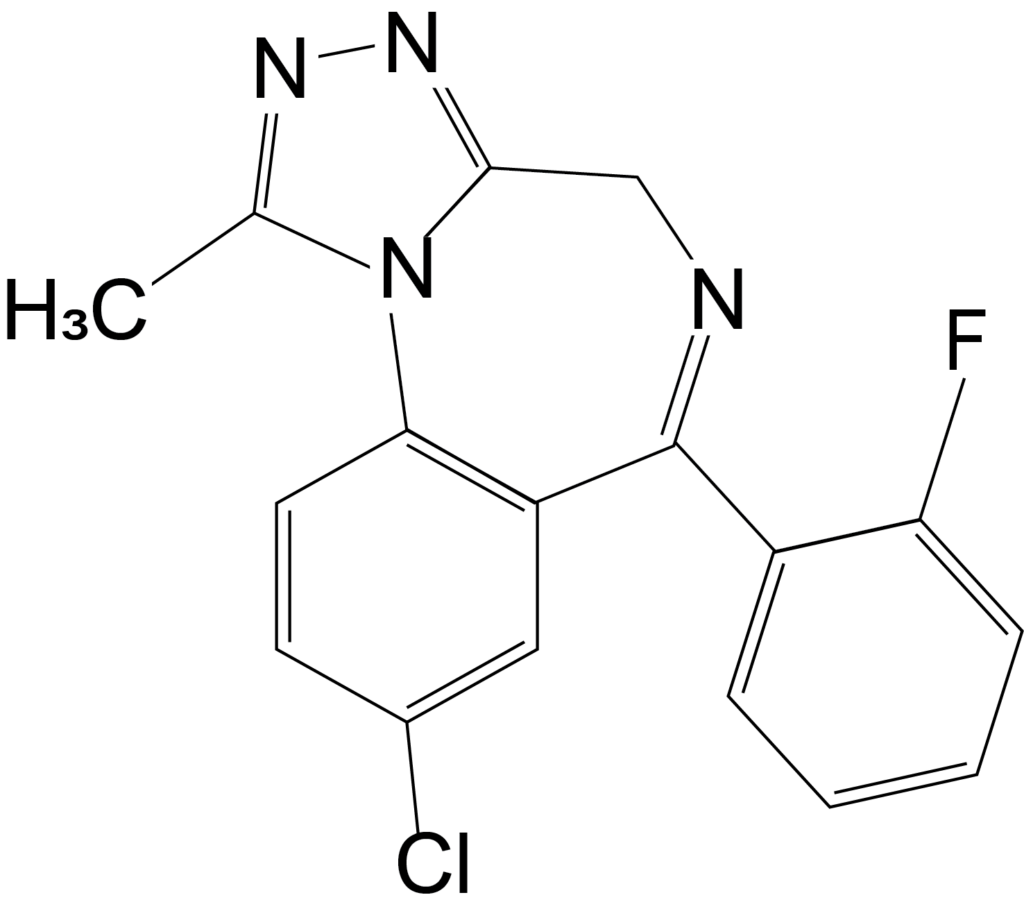 Flualprazolam is another such designer benzodiazepine which is not approved for use in the United States. Less is known about the potency of flualprazolam, but it is thought to create severe sedation and coma2. Flualprazolam is thought to be misused for its sedative and hypnotic effects, and users report the effects are similar to clonazepam and alprazolam2. The DEA only reported one case involving flualprazolam in 2017, but there were 1,624 cases in 2019.
Flualprazolam is another such designer benzodiazepine which is not approved for use in the United States. Less is known about the potency of flualprazolam, but it is thought to create severe sedation and coma2. Flualprazolam is thought to be misused for its sedative and hypnotic effects, and users report the effects are similar to clonazepam and alprazolam2. The DEA only reported one case involving flualprazolam in 2017, but there were 1,624 cases in 2019.
The DEA released an intent to temporarily schedule these two designer benzos as Schedule 1 substances in December 20223.
References:
- ETIZOLAM (usdoj.gov)
- Flualprazolam (Street Name: Flualp) (usdoj.gov)
- Federal Register :: Schedules of Controlled Substances: Temporary Placement of Etizolam, Flualprazolam, Clonazolam, Flubromazolam, and Diclazepam in Schedule I
Learn more about Benzodiazepines Testing at USDTL.
- Forensic vs. Clinical Drug Testing: Why Flexibility Matters for Your Organization
- USDTL’s Integration and Partnership With CourtFact
- New Year, New Capabilities: Offering Forensic & Clinical Testing Options
- Weather Delay
- The Detection of Delta-9-tetrahydrocannabinol, Delta-8-tetrahydrocannabinol, Delta-10-tetrahydrocannabinol, and Cannabidiol in Hair Specimens
- Umbilical Cord Tissue Testing for Ketamine
- Drugs of Abuse: A DEA Resource Guide (2024)
- Beyond THC and CBD: Understanding New Cannabinoids
- February 2026 (1)
- January 2026 (3)
- October 2025 (1)
- July 2025 (3)
- May 2025 (2)
- April 2025 (2)


