White Papers
Ethyl Glucuronide (EtG) and Ethyl Sulfate (EtS) Concentrations Following Use of Ethanol Containing Hand Sanitizer
USDTL Research
By: Joseph T. Jones, Mary R. Jones, Charles A. Plate, Douglas Lewis
Published: December 6, 2006

By Canva© Studio
Introduction
Ethyl glucuronide (EtG) and ethyl sulfate (EtS) are minor nonoxidative direct biomarkers of ethanol ingestion. The 2-5 day detection window of EtG and EtS is superior to direct ethanol measurements, which is approximately 1 hour per standard drink. The sensitivity of EtG and EtS in urine for beverage ethanol consumption is greater than 90 percent and is therefore an extremely important assay for the early detection of relapse in alcohol abstention programs.1,2 Other indirect markers, such as CDT,
GGT, MCV, EDAC, WBAA, and FAEE have sensitivities of less than 90 percent3 and are therefore useless as a key indicator of relapse. The controversy outlined in the Substance Abuse Treatment Advisory4 raises the issue of the detection of EtG and EtS in urine as being too sensitive. To date, there is little published data on the correlation of unintentional ethanol exposure and the detection of EtG and EtS in urine. One possible source of unintentional ethanol ingestion is the use of ethanol-containing hand sanitizers. These products can contain between 60-65% ethanol by weight. A typical purse-sized package (4 fluid oz) contains as much as six standard 12g drinks of ethanol. Ethanol is readily absorbed into the skin.
Experimental
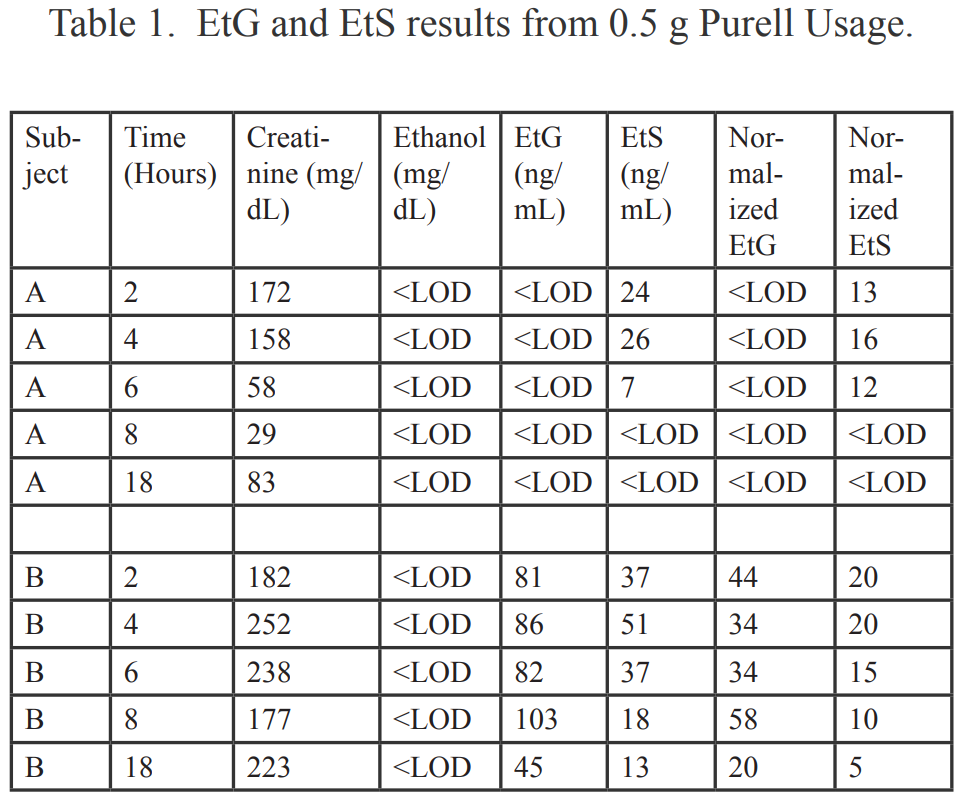
Dermal absorption of ethanol was studied by applying 0.5 g of Purell to the hands and messaging the gel into the hands. Purell, a popular over the counter anti-bacterial hand sanitizer, was chosen because of the large number of complaints in the general public that its use triggered positive urine EtG and EtS results. According to the product label, Purell contains 62% ethanol, which is an active antimicrobial ingredient. The equivalent number of 0.5 g doses to equal 1 standard 12g drink is 35-40 doses. This amount was considered to be adequate because it was just enough to entirely wet the hands up to the wrists. After both subjects demonstrated negative EtG and EtS baseline results, Purell usage was initiated and carried out once an hour for eight hours. Urine specimens were collected at 2 hours, 4 hours, 6 hours, 8 hours, and 18 hours (first void next morning). The specimens were analyzed at USDTL for creatinine and ethanol on an Olympus AU640 using standard protocols. The limit of detection for urine ethanol was 3 mg/dL. EtG and EtS determinations were performed at USDTL using standard protocols on an API 2000 LCMSMS. The limit of detection for EtG and EtS was 38.7 ng/mL and 7.2 ng/mL, respectively.
A second study using 2.0 g of Purell was conducted by Subject A. This amount was considered to be excessive because the subject had to rely on messaging the gel up to her elbows to thoroughly apply the excess gel. The equivalent number of 2.0 g doses to equal 1 standard 12g drink is 8-10 doses. This procedure was carried out once per hour for eight hours. Urine specimens were captured at 3 hours, 6 hours, 9 hours and 20 hours (first void next morning). The specimens were analyzed at USDTL for creatinine and ethanol on an Olympus AU640 using standard protocols. EtG and EtS determinations were performed at USDTL using standard protocols on an API 2000 LCMSMS. The limit of detection for EtG and EtS was 38.7 ng/mL and 7.2 ng/mL, respectively. Subject A noted that her hands and cuticles became irritated near the end of the study.
Study subjects
The two subjects that volunteered for this study are employees of USDTL. Subject A was a 64 kg 44-year-old white female. Subject B was a 127 kg 41-year-old white male. Both subjects are self-reported social drinkers who abstained from social drinking during study segments.
Results
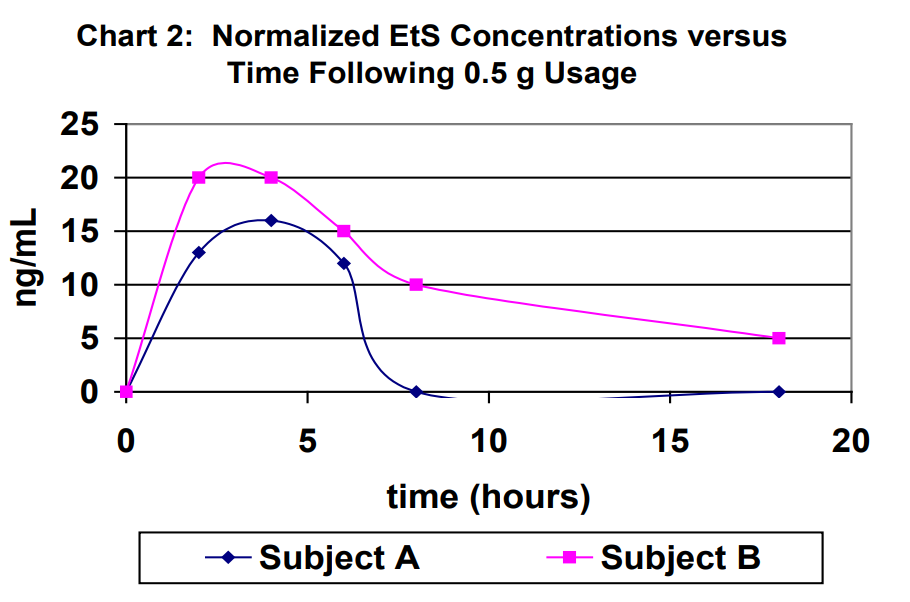
Urine specimens for each subject were captured at 2 hours, 4 hours, 6 hours, 8 hours and 18 hours (first void next morning) following 0.5 g usage of Purell. The results for the urine specimens are outlined in Table 1. For purposes of comparison, the results were normalized to a creatinine level of 100 mg/dL in Table 1. Normalized EtG results are shown in Chart 1. Normalized EtS results are shown in Chart 2.
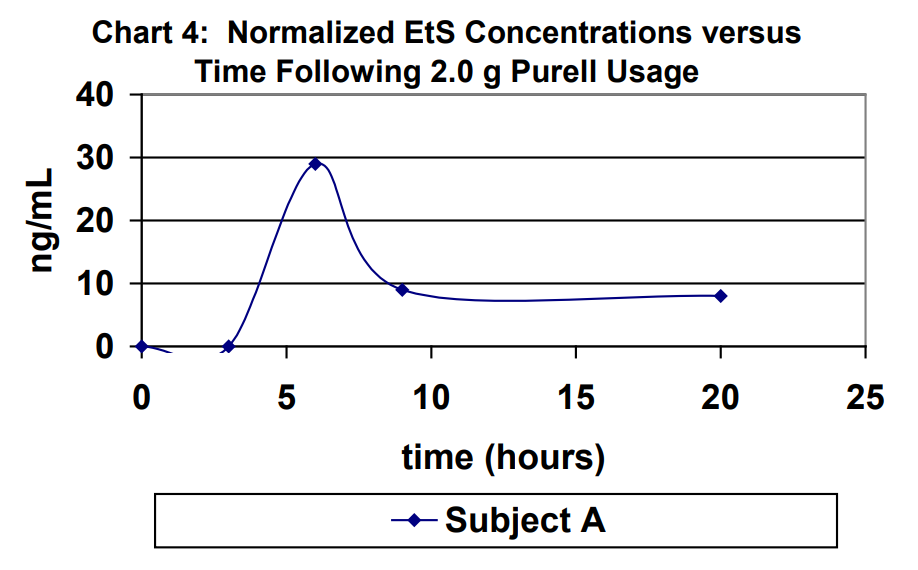
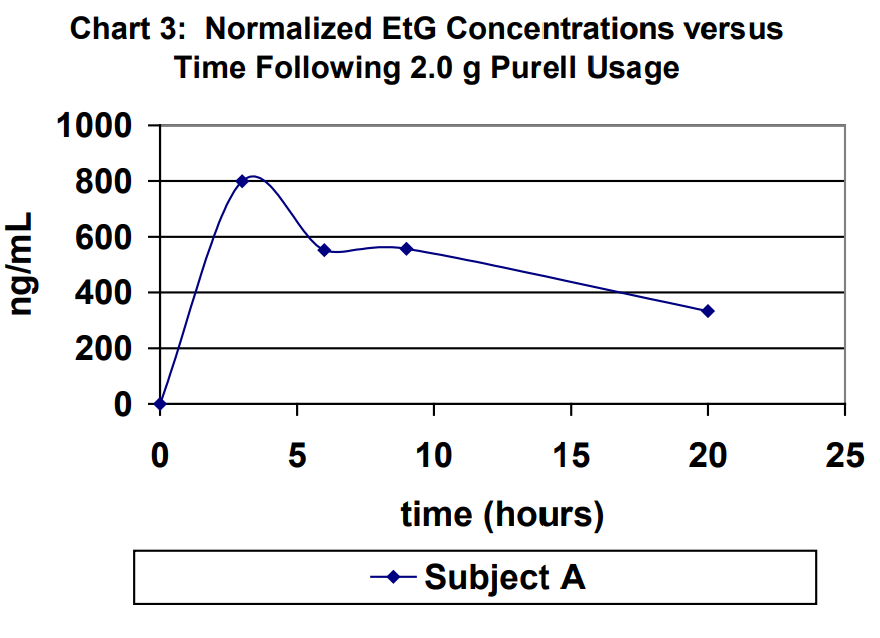
Urine specimens for Subject A were captured at 3 hours, 6 hours, 9 hours and 20 hours (first void next morning) following 2.0 g usage of Purell. The results for the urine specimens are outlined in Table 2. For purposes of comparison, the results were normalized to a creatinine level of 100 mg/dL in Table 1. Normalized EtG results are shown in Chart 3. Normalized EtS results are shown in Chart 4.

Discussion
Ethyl glucuronide and ethyl sulfate are very sensitive direct biomarkers of ethanol intake, both intentional and unintentional. This study demonstrated that it is possible to generate detectable concentrations of EtG and EtS by using ethanol-containing hand sanitizers. The highest EtG and EtS concentration achieved using the ethanol containing hand sanitizer eight times over an eight-hour period of time of normal usage was 103 ng/mL and 51 ng/mL, respectively. The highest EtG and EtS concentration achieved using the ethanol containing hand sanitizer eight times over an eight-hour period of time of excessive usage was 713 ng/mL and 14 ng/mL, respectively. Therefore, it is imperative that individuals participating in a program that requires ethanol- abstinence to either choose a hand sanitizer that does not contain ethanol for their personal hygiene or use soap and water. On the other hand, use of hand sanitizers cannot be used as an explanation for concentrations that are significantly higher than those demonstrated in this study.
References
- Wurst F; Seidl S; Ladewig D; Muller-Spahn F; Alt A. Ethyl Glucuronide: on the Time Course of Excretion in Urine During Detoxification. Addiction Biology 2002, 7, 427-434.
- Helander A; Beck O. Ethyl Sulfate: a Metabolite of Ethanol in Humans and a Potential Biomarker of Acute Alcohol Intake. J Anal Tox 2006, 29, 270-274.
- Bean P. State of the Art: Contemporary Biomarkers of Alcohol Consumption. Med Lab Obs. November, 2005.
- Center for Substance Abuse Treatment. The Role of Biomarkers in the Treatment of Alcohol Use Disorders. Substance Abuse Treatment Advisory. DHHS Publication No. 06-4223. Volume 5, Issue 4, September 2006.
Contact USDTL
1.800.235.2367
Client Services
Business Hours (CT)
Mon.-Fri. 7am-6pm
Sat. 8am-5pm
Holidays Closure
New Year's Day
Memorial Day
Independence Day
Labor Day
Thanksgiving Day
Christmas Day
Request Your Collection Supplies
For your convenience, USDTL provides test collection supplies at no additional charge.
Newsletters, Posters, and Catalogs
Our print materials will keep you up to date on the latest news in drug and alcohol testing.